Piv plugin imagej4/25/2024 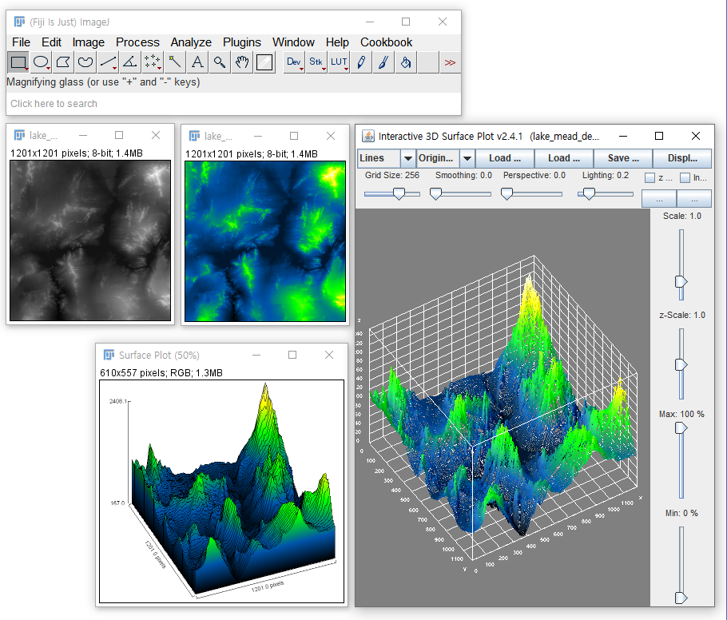
Generally, the mask should contain all possible localizations for your stains or dyes, or it should be a mask of localization for your null hypothesis ( i.e if you hypothesize a protein is localized to the mitochondria, you would want your mask to encompass the entire cell). This should be a mask of where within the image your want to search for spatial correlations. To get the best possible results, you will want to create and save a segmented mask for one of your images. Prepare a mask for the pixel randomizationĪnalysis mask: An example of an appropriate mask for analyzing cross-correlation of cytoplasmic proteins(right), generated from an actin stain (left). Verify accurate scaling (Image > Properties).Measure and subtract the mean background for both images (Process > Math > Subtract).Convert images to 32-bit (Image > Type > 32-bit).Deconvolve the images (Optional but recommended).Prepare a mask of the region to be analyzed.How to use Colocalization by Cross Correlation (CCC): Brief overview 
Installing the Plugin:Īvailable on the list of ImageJ updates sites. It currently works on 2D/3D single-channel images, supports time-series analysis, and requires a mask of all possible localizations for the signal in one of the images. It does this by performing a cross-correlation function (CCF) between the two images, operating in a similar manner to Van Steensel’s CCF except this plugin performs the CCF in all directions and provides additional information, such as the standard deviation and statistical measures. As a first step, a simple GUI has been introduced, with a workflow that resembles JPIV.This plugin attempts to determine: the average distance between non-randomly spatially associated particles, the standard deviation of that distance (which should also reflect the width of the PSF in the image for diffraction limited images), and two statistical measures of the association. 
In 2020, a collaboration with the OpenPIV project has started. Since 2004, JPIV is in use for research and education and constantly maintained. I developed JPIV in the scope of the work on my PhD thesis about micro-scale blood flow measurements. soil deformation when a pile is driven into the ground.movement of podosomes (structures on animal cell membranes).red blood cell aggregation in microchannels.Quick Start Guide to JPIV for Beginners.Ĭheck out the creative output of JPIV users on Google Scholar.If you experience an out of memory error or poor performance, increase the avalable memory: To allow up to four gigabytes, type java -jar -Xmx4G JPIV.jar Open a terminal and cd into the new directory.Prerequisite: A Java Runtime Environment (JRE). batch processing of a list of files as well as directories.velocity profile extraction, flow estimation.vector field graphics export (eps, pdf, emf, svg, png, jpeg.image display (png, tif, pgm, imx, im7).calculation of vorticity and other derivatives.rectangular interrogation areas possible.vector field filtering, outlier detection.ensemble correlation / ensemble averaging.multi-pass, multi-grid, FFT based correlation algorithm.JPIV is widely used in many fields of research.Ĭheck out the Quick Start Video on Vimeo! JPIV is written in Java and thus not rely on a special operating environment or Matlab although you can integrate scripts into the JPIV workflow that are written in Matlab, Octave, Python or other languages. JPIV is a complete, open source, and platform independent software for 2D Particle Image Velocimetry (PIV).
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
